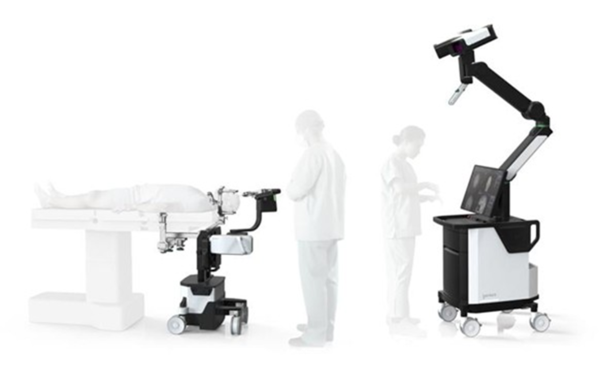
Koh Young Technology said on Tuesday its brain surgery medical robot, Geniant Cranial, has obtained approval from Japan’s Pharmaceuticals and Medical Devices Agency (PMDA). The approval allows Koh Young to sell the robot officially in Japan.
Geniant Cranial, a Koh Young robot, secured U.S. Food and Drug Administration 510(k) clearance in January last year and has now obtained approval from Japan’s PMDA as a high-risk Class III medical device. With this, Koh Young becomes the first South Korean robotics company to secure approvals from both the U.S. FDA and Japan’s PMDA in the brain surgery field.
Japan is seeing a rapid rise in brain diseases and degenerative neurological disorders as it enters a super-aged society. Demand is also continuing to expand for minimally invasive brain surgery that can raise precision while reducing the burden on patients. About 1,700 neurosurgery hospitals capable of brain surgery operate across Japan.
Geniant Cranial is used in various neurosurgical areas, including epilepsy, movement disorders such as Parkinson’s disease, and brain tumor biopsy procedures. Its surgical guide robot and navigation support accurate position matching and real-time tracking. It is designed to provide medical staff with a precise and intuitive surgical environment.
A Koh Young Technology official said, "Japan has strong demand and high interest among medical staff in precision robot surgery, so inquiries from leading neurosurgery professors have continued even before approval." The official added, "Through our Japan unit, we will quickly push cooperation with major hospitals and early installations to expand our surgical robot business in Japan in earnest."
